Contents
Let’s look at what covalent bonds are, how they form, and their characteristics, with easy examples.
Let’s Review the Atom
First, let’s remember that atoms are made of protons, neutrons, and electrons.
The last ones, electrons, have a negative charge and are the ones that come into play in covalent bonds. Therefore, they are the only ones we are interested in for this topic.
Electrons spin in orbits around the atom’s nucleus, called orbitals, shells, or energy levels.
Also, remember that atoms can join together to form what is called a chemical bond. These chemical bonds are precisely the forces that hold atoms together.
How Do Atoms Bond?
One of the ways they bond is by forming covalent bonds, which are what we will explain here.
When two atoms bond, they give, take, or share electrons. But only the so-called “valence electrons” can do this—be given, taken, or shared by another atom.
Valence electrons are those found in the outermost shell of the atom, and they are the only ones available to be shared with another atom.
When two atoms bond, they always follow the so-called octet rule.
The Octet Rule
The octet rule, also called Lewis’s law, states that all atoms of the elements in the periodic system tend to complete their outermost energy levels (shells) with a total of 8 electrons.
It’s the electrons in the outermost shell, those farthest from the nucleus, that tend to be completed to a total of 8. To do this, they will share electrons with another atom if necessary.
NOTE: The noble gases are the only non-reactive elements (they don’t react or share with anyone) because they already have 8 electrons in their outermost shell. They do not share any more.
This combination of electrons from the outermost shell between atoms can happen in several ways, one of which is the covalent bond.
The Covalent Bond
Covalent bonds are formed between NON-METAL atoms.
Remember a bond (atoms that share electrons and are therefore linked).
The elements on the far right of the periodic table are the non-metallic elements (yellow color). Only these can form covalent bonds.
Non-metal atoms usually have many electrons spinning in their outermost orbit (called valence electrons), so they tend to gain (take) electrons from another atom rather than giving them up to have the 8 electrons required by the octet rule and achieve the stability of the noble gases (having 8 valence electrons).
The problem is that since both atoms are in this situation—both being non-metals—neither one will want to give up its electrons to the other.
What does this mean?
Well, these atoms, the non-metals, since they don’t want to lose electrons, will share electrons from their outermost shell when they meet or bond, instead of giving or gaining them, which would be the case for ionic bonds.
Conclusion: Non-metallic atoms cannot give or gain electrons among themselves; instead, they share them.
“Covalent bonds form between two nonmetals by sharing valence electrons.”
When two non-metallic atoms bond, the electrons they share hold them together and become part of both atoms, thus forming a molecule (several bonded atoms). Once bonded, both atoms acquire the structure of the noble gases with 8 electrons.
They can be joined by single, double, or triple bonds, depending on the electrons they share in the union.
Formation of Covalent Bonds
Let’s see an example of covalent bond formation.
Chlorine is a NON-METAL. In its natural state, chlorine exists as Cl₂, meaning a chlorine molecule of 2 atoms. The two chlorine atoms are joined by a covalent bond.
Chlorine has 7 electrons in its outermost shell, so if they each share one of these electrons, they would each have 8 electrons in the molecule.
Perfect, they have formed a very stable molecule with two atoms. This bond only needs to share one electron from each atom to form the octet.
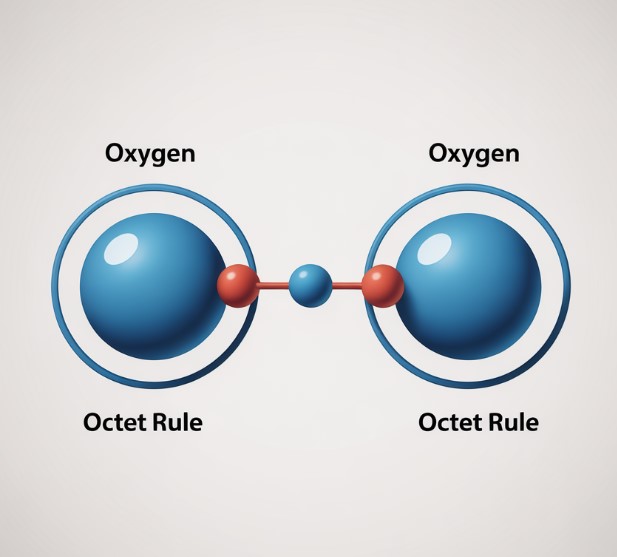
Another case is the oxygen molecule, O₂. Each oxygen atom has 6 valence electrons, so they will have to share 2 electrons each to reach the octet.
Covalent bonds can share 1, 2, or 3 valence electrons. They are called single, double, or triple bonds. Here is an example of each:Cl₂, O₂, and N₂.
Normally, these bonds are written by placing a small circle for each valence electron of each atom and a line for each of the shared electrons in the bond.
F₂ shares 1 electron, so one line. O₂ shares 2 electrons, so 2 lines. The first is a single bond, and the second is a double bond. As you know, there are even triple bonds.
The question is, how do I know how many valence electrons any given atom has?
This is called the atom’s electronic configuration, and you can find it by going to the following link: Electronic Configuration. Another simple way is to know the families of the periodic table, which will also tell you the valence electrons.
If we know the electronic configuration of the non-metals that form the covalent bond, it’s very easy to see what the covalent bond looks like.
The periodic table of elements sometimes also specifies the number of valence electrons for each atom.
Let’s give you an exercise. Don’t worry, we’ll give you the solution step-by-step.
Determine if the bond formed in the following covalent unions is single, double, or triple.
N₂, I₂, O₂, Br₂
Solution:
N₂ = Nitrogen has 5 valence electrons. Since all atoms seek the most stable valence state, which is the octet, it will form a triple bond.
I₂ = A single bond because they have 7 valence electrons.
H₂ = A single bond. Hydrogen is an exception to the octet rule; it only seeks to have two electrons in its outermost energy level.
O₂ has a double bond since it has 6 valence electrons.
Br₂ = Forms a single bond since it has 7 valence electrons.
So far, we’ve covered the simplest covalent bonds, but there are slightly more complex ones.
Let’s look at a bond of 3 nonmetals: CO₂, or carbon dioxide. It has the following expression: O=C=O. Why?
Oxygen has 6 valence electrons, and carbon has 4. How do they form the covalent bond? Well, the carbon shares its 4 valence electrons with the other two oxygen atoms, so the oxygen atoms have 8 electrons. This is a bit more difficult.
This type of bond is called a coordinated or dative covalent bond: it is a covalent bond in which the shared pair of electrons is provided by a single atom (in our case, carbon). Compounds with this type of bond are called complexes.
To form this type of bond, you need to know the so-called Lewis structure, which is an interpretation of covalence.
Electronegativity is the ability to attract electrons.
In the periodic table, electronegativity decreases from top to bottom within a group, while it increases from left to right across a period.
Additionally, there is an electronegativity table with specific electronegativity numbers for each atom.
Another example is the water molecule, H₂O.
An oxygen atom has six electrons in its outermost shell, so it needs two more to complete it. That’s why it bonds with two hydrogen atoms, each of which contributes its single electron.
The result is the formation of a water molecule in which there are two oxygen-hydrogen covalent bonds.
This is a molecule with a central atom, in this case, carbon, in CH₄ (methane). This atom has 4 valence electrons that it will share, one with each hydrogen atom, to achieve a stable configuration, forming an electron cloud shared by the molecule.
Examples of Covalent Bonds
Water molecule (H₂O), ammonia (NH₃), H₂ (dihydrogen gas), O₂ (oxygen gas), Cl₂ (chlorine gas), Br₂ (elemental bromine), N₂ (nitrogen gas), CH₄ (methane), C₂H₆ (ethane), S₈ (rhombic sulfur), P₄ (white phosphorus), and NF₃ (nitrogen trifluoride).
Characteristics of Covalent Bonds
- Covalent compounds can exist in any state of matter: solid, liquid, or gas.
- They are poor conductors of heat and electricity.
- They have relatively low melting and boiling points.
- They are soluble in nonpolar solvents like benzene, carbon tetrachloride, etc., and insoluble in polar solvents like water.
If you liked this, please click Share. Thanks.